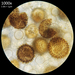
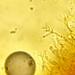
Photos / Sounds
What
Amorphous Bird's Nest Fungus (Nidularia deformis)Observer
jjjjaaaammmmiiiieeeePhotos / Sounds
What
Shelf Fungi (Order Polyporales)Observer
lostcoastmikeDescription
Too early to find spores.
Photos / Sounds
What
Eyed Beard (Usnea quasirigida)Observer
wweellllDescription
quasirigida continuing its march south
Photos / Sounds
What
Deer-colored Trametes (Trametopsis cervina)Observer
lostcoastmikeDescription
Spores 6-7 X 2.5-3 µm.
My first ones!
Photos / Sounds
What
Ciboria rufofuscaObserver
kendrasveaDescription
Doug fir cone. Seemed like some smaller ascos growing off the larger Ciboria - curious if this is common, and if it's the same species.
Photos / Sounds
What
Earth Tongues (Genus Geoglossum)Observer
lostcoastmikeDescription
No hairs.
Spores 79-85 X 5.5-6 µm.
Asci 180 x 20 µm.
Photos / Sounds
What
Xylodon quercinusObserver
lostcoastmikeDescription
Spores 5-6 X 3-3.2 µm.
Maybe Sistotrema. . . .
Photos / Sounds
What
Ceriporia purpureaObserver
lostcoastmikeDescription
Maybe. . . based on spore shape.
Spores 5 X 1.5 µm
Photos / Sounds
What
Rose-petalled Jellyskin Lichen (Scytinium gelatinosum)Observer
rambryumDescription
On Eastern Cottontail Fecal Pellets --aged
Photos / Sounds
What
Artist's Conk (Ganoderma brownii)Observer
alan_rockefellerDescription
On Quercus agrifolia
Spores measure (10.8) 11.3 - 13.2 (13.5) × (6.2) 7 - 8.2 (8.5) µm
Q = (1.4) 1.44 - 1.7 (1.8) ; N = 30
Me = 12.1 × 7.6 µm ; Qe = 1.6
Photos / Sounds
What
Boletes and Allies (Order Boletales)Observer
jackjohnsonnDescription
Leucogyrophana, Serpula?
Photos / Sounds
What
Giant Gray Milk Cap (Lactarius argillaceifolius var. megacarpus)Observer
alan_rockefellerDescription
Very spicy after 15 seconds
Photos / Sounds
Observer
lostcoastmikeDescription
Steccherinum ochracea have smooth spores, these are ornamented with warts.
Photos / Sounds
What
Mycena robustaObserver
drdick98229Description
Photomicrographs 1, 2 and 3 are cheilocystidia; 4 and 5 are of the pileipellis.
Photos / Sounds
What
Shiitake (Lentinula edodes)Observer
lostcoastmikeDescription
An escapee on downed Tanoak tree, wildly!
Same tree as last season.
Photos / Sounds
Observer
hendre17Description
Fruiting in mossy patch of grass near the state Capitol building campus.
Cap: orange/striate. Hygrophanous(drying pale buff to nearly white).
Stem: heavily pruinose/frosted(especially at apex). Slightly darker/grayish at base.
Harvested 5 specimens.
Spore printed a single cap directly on a glass slide.
Spore print: Pale rusty orange.
Mounted spore printed slide in Lugol’s solution to test for Dextrinoid reaction.
Spores: Ovoid to amygdaliform, lumpy/roughened.
NON-DEXTRINOID in Lugol’s solution.
Medium sized.
Thoroughly dehydrated specimens and bagged for herbarium collection/genetic record.
My coinciding Mushroomobserver observation below-
Photos / Sounds
What
Layered Cup (Peziza varia)Observer
lostcoastmikeDescription
Spores 12-15 X 8-10 µm.
Asci tips amyloyd in Mezler's.
Photos / Sounds
What
Blood Lichen (Genus Mycoblastus)Observer
sadie_hickeyDescription
On conifer branch stub
Photos / Sounds
What
Candy Cap (Lactarius rubidus)Observer
hendre17Description
Fruiting near heavily cubical rotted log/debris beneath 2nd growth Douglas fir and maple.
Odor: pungent sugar cookie odor in the field. Intensifying to maple syrup odor after hours of dehydration.
Cap: dry, lumpy, Kevlar like.
Stem: very brittle/hollow.
Latex: watery, cloudy grey, skim milk.
I took two friends here today and shared this patch with them/presented on ecology, description/features/look alikes/etc.
I gave the majority of harvested specimens to them to keep. I kept a few for myself. Rapidly dehydrated a single specimen in dehydrator for herbarium collection/genetic record with this observation.
My coinciding Mushroomobserver observation below-
Photos / Sounds
What
Cannonball Fungus (Sphaerobolus stellatus)Observer
jackjohnsonnDescription
on woody debris in Doug fir dominant area, some Picea sitchensis present.
PCR done by @mgkoons
Photos / Sounds
Observer
hendre17Description
Fruiting beneath Sitka Spruce and Western Hemlock.
Completely viscid top to bottom.
Harvested both specimens.
Identified correctly by Noah Siegel.
Dehydrated both specimens and bagged for herbarium collection/genetic record.
My coinciding Mushroomobserver observation below-
Photos / Sounds
What
Deadly Webcap (Cortinarius rubellus)Observer
dmallochDescription
Malloch 29-08-23/01
Solitary at the side of a path among Dicranum sp. in a pure stand of Abies balsamea
In the herbarium of the New Brunswick Museum (NBM)
Basidiospores orange brown in spore print, ovate in profile, coarsely roughened but often discharged in an apparently immature and nearly smooth state, not strongly dextrinoid, 8.9-11.6 x 6.1-7.6 µm, Q = 1.31-1.88 (average[49]: 9.8 ± 0.5 x 6.7 ± 0.4 µm, Q = 1.47 ± 0.10)
Cheilocystidia resembling immature basidia, clavate, 10.1-16.6 x 3.5-5.0 µm
Pileipellis a loose cutis of repent to subascending encrusted red brown hyphae, with a conspicuous nearly brick-like hypoderm of short broad hyphae
Photos:
- Basidioma
- Cross section of pileipellis in Windex
- Cheilocystidia in KOH + Congo Red
- Encrusted hyphae of the lamellar trama in KOH + Congo Red
- Cross section of lamella in KOH showing cheilocystidia
- Basidiospores from spore print in Melzer's Solution
Photos / Sounds
What
Funeral Bell (Galerina marginata)Observer
jackjohnsonnDescription
On large fallen tree- interesting blue fluorescence in UV
Photos / Sounds
What
Candy Cap (Lactarius rubidus)Observer
hendre17Description
Took Boyd’s good friend Roxanne and her folks to our candy cap patch and gave them a tutorial/short presentation on identifying features, look alikes, etc.
All specimens presenting thin/watery latex(skim milk).
Odor: strong sugar cookie dough(in the field).
Quabalactone 3 converting to sotolon after 3hrs(to 4 days) of dehydration. I always dehydrate them in dehydrator for 2-3hrs at 90F to ensure they won’t mildew. Then let them air-dry slowly on racks in our house for remaining 2-3 days until thoroughly dry.
Harvested multiple specimens and dehydrated for later culinary use.
Bagged a single specimen for herbarium collection/genetic record.
My coinciding Mushroomobserver observation below-
Photos / Sounds
What
Umber-brown Puffball (Lycoperdon umbrinum)Observer
lostcoastmikeDescription
Spores 4.5-5 µm Diam.
Photos / Sounds
What
Candy Cap (Lactarius rubidus)Observer
hendre17Description
Fruiting abundantly beneath 2nd growth Douglas fir and very young Western Hemlock saplings(amidst multiple older stumps and cubical rotted wood).
Latex: watery, clear, skim milk(not white).
Odor: sweet/sugar cookie in the field. Once home/on drying racks becoming intense with maple syrup/currry scent.
Slowly drying multiple specimens for later culinary use. Dehydrated a single specimen in higher heat/dehydrator for herbarium collection/genetic record.
My coinciding Mushroomobserver observation below-
Photos / Sounds
What
Bowl Hoodie (Calyptella capula)Observer
lostcoastmikeDescription
On old Dill Stalks, Anethum graveolens.
Photos / Sounds
What
Inocybe curvipesObserver
hendre17Description
Fruiting in soil/moss on the edge of a clearing. Douglas fir dominant in this area but numerous young Alnus rubra saplings were within a few feet of this flush. Initially mistook this as a Telamonia and labeled my voucher sheet as such. Changed it to Inocybe once noting the spermatic odor, vertically striate/pruinose stem, pale gray brown spore print and spore shape.
Cap: distinctly papillate, acutely umbonate with many young specimens, densely scaly between disc and margin. Deep chestnut/ferruginous brown. White margin band.
Stem: vertically striate, slightly pruinose towards apex, rather equal(not bulbous at base).
Harvested 7 specimens.
Odor: indistinct in the field. Once home spermatic odor detected(strong near stem apex where cut).
Spore printed two caps on two slides(one in each slide).
Spore print: grayish brown/straw brown.
Mounted printed slide in KOH.
Spores: trapeziform, angular, faintly nodulose with an elongated node at apex(pointed snout). 6-8 nodes in most angular spores. Some irregularly curved.
Mounted a third slide with a single removed lamella. Analyzed gill edge.
Cystidia ranging from very small balloon shaped to long Metuloid appearing cystidia with granular apex.
Dehydrated all specimens and bagged for herbarium collection/genetic record.
My coinciding Mushroomobserver observation below-
Photos / Sounds
What
Small-warted Mountain Puffball (Lycoperdon subcretaceum)Observer
lostcoastmikeDescription
Spores (4) 5-6 µm Diam.
Photos / Sounds
What
Fungi Including Lichens (Kingdom Fungi)Observer
mycomutantPlace
PrivateDescription
Structures found in agar culture of Leucocoprinus birnbaumii from potting soil. The agar has sclerotia present from L. birnbaumii and these tiny spheres were found in the mycelium and agar amongst them or clinging to the sclerotia.
Observation of the sclerotia: https://www.inaturalist.org/observations/185223484
Macroscopically the agar had reddish brown areas and under 40x magnification was filled with brown specks such that it appeared like tapioca - and had a similar consistency. The agar was like slime and became a tangled mess if attempts were made to isolate any of these structures, besides the sclerotia which were large enough to remove. As such pieces of the slimy agar were simply crushed beneath a cover slip to study.
Some of the structures are brownish, spiky balls ~ 35-45 µm in diameter. Also some similar looking ones that were whitish or clear which could be immature ones.
Additionally smaller spore like structures were present that were 7 - 9 µm in diameter. These were smooth, roughly spherical and appeared hyaline though colour was hard to judge due to the agar. When Melzer's reagent was applied they developed a yellow colour with a thick, pinkish wall.
Additional spherical structures were present that were between the size of these two structures and around 20 µm in diameter. These were smooth, hyaline and yellow in Melzers but with a greenish wall.
I don't know what I am looking at here and whether some of these are chlamydospores, blastospores or something else. Besides the sclerotia that are from L. birnbaumii it is not clear what any of the other things present, including some of the mycelium, belongs to.
Photos / Sounds
What
Frothy Porecrust (Irpex latemarginatus)Observer
kendrasveaDescription
Really unsure about this one - speculative ID. Particularly interesting how this had also taken over fern. Not slime mold, relatively firm. Rosette like form. Might go back to get this for sequencing. Primarily on alder (or birch?)
Photos / Sounds
What
Onion-stalk Parasol (Leucocoprinus cepistipes)Observer
mycomutantDescription
Spores: (6) 7-14 (15) x (5) 6-8 (9) µm. Ellipsoid, amygaldiform or subglobose. Hyaline, smooth and with a large greenish oil droplet/guttule inside and a capped germ pore. Dextrinoid, congophilous, cyanophilous, metachromatic in Cresyl blue with pinkish purple inner wall. Very slight pinkish colour in 1% Aniline blue in lactic acid. Pinkish colour to inner spore and yellowish tone to wall observed in 5% KOH mount becoming more noticeable in 10%.
Guttules were mostly single, some double or triple. The largest single ones observed measured: 3-4 x 3-4.5 µm.
Dried mushroom cap showed a very slight pinkish tone when 5% KOH was applied, though mostly just bleached. 10% FeSO4 resulted in similar bleaching but no colouration was noted.
Leucocoprinus species sent to me from a Reddit user in the UK. Growing in a potted Aspidistra plant inside. Presumably Aspidistra elatior.
https://www.reddit.com/r/mycology/comments/151e3sn/id_for_these_fellas_uk/
Spore prints and a number of dried specimens were received - the gills of which had discoloured to such a greenish grey colour that they initially looked like they could be moldy, since they had rehydrated slightly in the mail. Upon closer inspection however this was just discolouration, or possibly was the result of the greenish guttules/oil droplets inside the spores. The spore print appeared whitish on the black paper but the slides with gill sections mounted showed a slightly greenish grey colour where spores were spilling out, visible with the naked eye. So perhaps enough spore mass in a print would have the same tone.
Macroscopically the mushrooms appear similar to Leucocoprinus cepistipes with the distinctive looking shape of the immature mushrooms complete with the clear exudation.
I have seen a similar greenish grey gill discolouration in observations before, see here:
https://www.inaturalist.org/observations/20082876
https://www.inaturalist.org/observations/32393475
Whereas other times a more reddish pink, brownish or yellowish tone is present. I'm not clear if there is any distinction between these or if it just represents different stages of discolouration.
Microscopically however the spores were far larger than those noted in descriptions of L. cepistipes. A size of (6) 7-14 (15) x (5) 6-8 (9) µm was observed from spores spilling out of a gill section crushed under the cover glass. From the spore print however a smaller size was observed of 7-11 x 6-7 µm. The larger spores were immediately obvious upon looking at a gill section and were not just outliers.
Flora Agaricina Neerlandica vol. 5 gives a spore size for L. cepistipes of 7.5 - 13 x 6-8 µm so this would seem out of that range.
I have also observed this with L. ianthinus where a gill section results in a larger spore size reading than from the print. So it appears that measuring the spore size of Leucocoprinus species from prints may not be reliable. I have not read anything to the effect of spore prints being less reliable than gill section mounts for measuring spore size. Mushroom Expert seems to note the opposite, mentioning that immature spores may be seen with a gill mount. Spores that appeared obviously immature and undeveloped weren't included in my measurements, but seemed fewer than the large spores anyway. Most of the spores with a measurment of 5µm were closer to spherical and either 5x5 or 6x5. They appeared fully formed though rather than immature.
I am speculating that perhaps Leucocoprinus species retain more of the large spores when they are found outside of high humidity, tropical environments as a larger buller's drop would need to form in order to release larger spores. However I've never looked for this phenomenon in common, native garden mushrooms so will have to see if I record the same thing there.
Comparing the photos of the mushrooms and the size of the spores to the 80 odd species of Leucocoprinus for which I have adequate information I cannot find a direct match. My best guess currently is that this may be something that has been described as a variant or form of L. cepistipes. So I'm going to try and get the description for Leucocoprinus cepistipes f. macrosporus Migl. to see if this may match and explain the larger spores.
They appear similar to this observation from New Zealand, also found in an Aspidistra plant so perhaps the plant may point to an origin for this species/variant. Will need to look for more observations with these plants.
Photos / Sounds
What
Red-pored Bolete (Rubroboletus pulcherrimus)Observer
hendre17Description
Very young fruiting bodies on the edge of 3rd beach beneath Sitka Spruce and Western Hemlock. Only a few feet from this observation= https://mushroomobserver.org/527063?q=1p1WK
Photographed on my way back up the trail from 3rd beach.
Left all young specimens to mature.
Photos / Sounds
Observer
rambryumDescription
Best guess for dark muriform spores in perithecia on the liverwort Marsupella in the alpine.
Photos / Sounds
Observer
profiniteDescription
Abundant under top layer
Scent: Garlicky garbage bin
Abies, Tsuga forest (rarely Pinus)
UV365-, faint on peridium crack
Photos / Sounds
What
Eyelash Cups (Genus Scutellinia)Observer
lostcoastmikeDescription
Spores~22 x 13 µ
Long Asci 1270-1320
Photos / Sounds
What
Pediastrum duplexObserver
maricel-patinoDescription
Found on the roots of aquatic plant that was growing in a seepage.
What
Antistrophus rufusObserver
nfurlanDescription
Reared from cryptic stem galls on S. laciniatum: https://www.inaturalist.org/observations/143918334
What
Antistrophus minorObserver
nfurlanDescription
Reared from cryptic stem galls on S. laciniatum: https://www.inaturalist.org/observations/143918334
Photos / Sounds
What
Fungi Including Lichens (Kingdom Fungi)Observer
lostcoastmikeDescription
Spores 5.5-7 X 4 µm.
Photos / Sounds
Observer
lostcoastmikeDescription
Spores (not spores, sclerotia: 23-25 x 20-24 µm.
Yellow in KOH.
Photos / Sounds
What
Genus PorellaObserver
lostcoastmikeDescription
@rambryum Could you help me with this ID? It might help me ID the slime mold on there too.
Thanks!
Spores, ? , 18-20 X 11-13.5 µ
Photos / Sounds
What
Cordyceps farinosaObserver
mgkoonsDescription
cf. found in the small cleared area in the lower center of the environmental context picture on the trails edge where the soil is wet but not muddy. On Lepidoptera cocoons in which the larvae are wrapped in a leaf fragment to transform before succumbing to this cordyceps sp
Photos / Sounds
What
White Inside-out Flower (Vancouveria hexandra)Observer
hendre17Description
Flowering.
Bob Bammert grove trail.
Shot with iPhone11.
Photos / Sounds
What
Charcoal Cup (Peziza echinospora)Observer
lostcoastmikeDescription
Spores 12.5-13 X 6-7 µm.
Photos / Sounds
What
Lachnum gaultheriaeObserver
corndogDescription
cf.
On Gaultheria, always causing circular leaf spots which later excise themselves from the the leaf and fall to the ground.
Photos / Sounds
What
Capitotricha bicolorObserver
lostcoastmikeDescription
Spores 9-11 X 2-2.5 µm.
Asci 58-65 x 5 µ.
Paraphyses: 90-110 x 5 µ, pointy tipped
Photos / Sounds
Observer
ryan_durandDescription
Nivicolous. Growing on dead, firm conifer wood on edge of melting snowbank.
Stalk 2.2mm, sporocarp 2.9mm, cylindrical with base wider than apex.
Peridium rupturing into small flakes.
Columella extending to just short of apex.
Capillitium dense, dark net, common nodes, small periphery spines.
Spores brown, 8.9-10.2, minutely warted.
Photos / Sounds
What
Merismodes anomalaObserver
lostcoastmikeDescription
Spores 9-10 X 4.2-5 µm.
Abundant clamps.
Photos / Sounds
What
Basidiomycete Fungi (Phylum Basidiomycota)Observer
lostcoastmikeDescription
Spores 6-7 X 5.5-6 µm.
Micro does not fit Dendrothele
Photos / Sounds
What
Arrhenia peltigerinaObserver
corndogDescription
On Peltigera in interdunal wetland
Photos / Sounds
What
Trametes pubescensObserver
jackjohnsonnDescription
I guess? Soft and somewhat rubbery weakly zonate hirsute unicolorous cap with angular pores which are elongated towards the margin to become Mazelike. On standing dead Alnus rubra in overlapping shingles. Whitish ochre pores that became slightly darker when rubbed.
Photos / Sounds
What
Genus GibellulaObserver
jackjohnsonnDescription
On large fallen decorticated tree in stream.